Successful management of intralenticular Ozurdex injection causing cataract and intractable glaucoma
Abstract
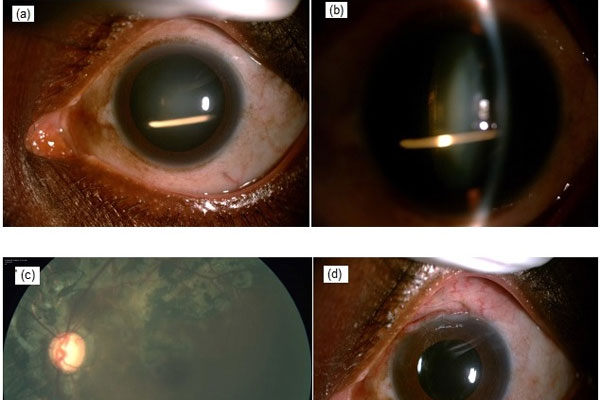
A 50-year-old female presented with profound vision loss and was previously advised injection Ozurdex in the left eye. In the left eye, the anterior chamber was quiet, intraocular pressure (IOP) was 58 mmHg and cataractous changes were noted with Ozurdex implant inside the lens substance. The left eye had glaucomatous cupping (0.9:1 CDR), bipolar rim thinning, inferior notch and healed choroiditis. The patient underwent phacoemulsification, trabeculectomy and mitomycin-C in the left eye. Visual acuity improved and IOP was under control. Although Ozurdex is effective, there are reports of complications related to the drug and implantation procedure. This case highlights an uncommon complication of an uncontrolled, persistent steroid response leading to glaucomatous optic atrophy and profound vision loss due to an accidental intralenticular implantation of Ozurdex. Our case reinforces the need for caution about the decision regarding the judicious use of intravitreal steroids and employment of appropriate technique.
References
Ozurdex® (Allergan, Inc, Irvine, CA, USA). http://www.ozurdex.com/. Last accessed 15 January 2018.
Haller JA, Kuppermann BD, Blumenkranz MS, et al. Dexamethasone DDS phase II study group. Randomized controlled trial of an intravitreous dexamethasone drug delivery system in patients with diabetic macular edema. Arch Ophthalmol. 2010;128(3):289-296.
Myung JS, Aaker GD, Kiss S. Treatment of noninfectious posterior uveitis with dexamethasone intravitreal implant. Clin Ophthalmol. 2010;4:1423-1426.
Ghosn CR, Li Y, Orilla WC, et al. Treatment of experimental anterior and intermediate uveitis by a dexamethasone intravitreal implant. Invest Ophthalmol Vis Sci. 2011;52(6):2917-2923.
Sekeroglu MA, Anayol MA, Koc F, Tirhis H, Ozkan SS, Yilmazbas P. Intralenticular sustainedrelease dexamethasone implant: is it still effective on macular edema. Case Rep Ophthalmol. 2016;7(1):85-89.
Coca-Robinot J, Casco-Silva B, Armadβ-Maresca F, GarcÃa-MartÃnez J. Accidental injections of dexamethasone intravitreal implant (Ozurdex) into the crystalline lens. Eur J Ophthalmol. 2014;24:633-636.
Fasce F, Battaglia Parodi M, Knutsson KA, et al. Accidental injection of dexamethasone intravitreal implant in the crystalline lens. Acta Ophthalmol. 2014;92:e330, e331.
Ram J, Agarwal AK, Gupta A, Gupta A. Phacoemulsification and intraocular lens implantation after inadvertent intracapsular injection of intravitreal dexamethasone implant. BMJ Case Rep. 2012;12:2012.
Sheen OS, Gepstein R, Assia EI. Intralenticular Ozurdex implant: a case report. Clin Surg. 2017;2:1556.
Munteanu M, Rosca C. Repositioning and follow-up of intralenticular dexamethasone implant. J Cataract Refract Surg. 2013;39:1271-1274.
Roy R, Hegde S. Split Ozurdex implant: a caution. Can J Ophthalmol. 2013;48(1):e15, e16.
Chang-Lin JE, Attar M, Acheampong AA, et al. Pharmacokinetics and pharmacodynamics of a sustained-release dexamethasone intravitreal implant. Invest Ophthalmol Vis Sci. 2011;5;52(1):80-86.
Copyright (c) 2020 Kalpa Negiloni, Ronnie Jacob George, Shwetha Tripathi, Sridharan Sudharshan

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors who publish with this journal agree to the following terms:
- Authors retain copyright and grant the journal right of first publication, with the work twelve (12) months after publication simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgement of its initial publication in this journal.
- Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) prior to and during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work (See The Effect of Open Access).


